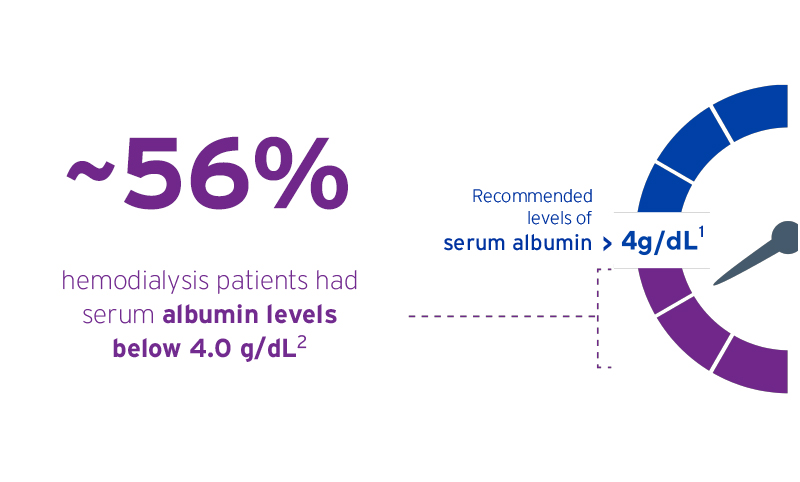
- NKF KDOQI Clinical Practice Guidelines. Am J Kidney Dis. 2000. Jun; 35(6 Suppl2}: S1-S140.
- United States Renal Data System. 2022 USRDS Annual Data Report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD, 2022.
- Don BR, Kaysen G. 2004. Serum Albumin: Relationship to Inflammation and Nutrition. Seminars in Dialysis 17(6):432-7.
- Tsuchida, Kenji et al. Albumin Loss Under the Use of the High-Performance Membrane, Department of Kidney Disease (Artificial Kidney and Kidney Transplantation), Kawashima Hospital, Tokushima City, Tokushima, Japan.
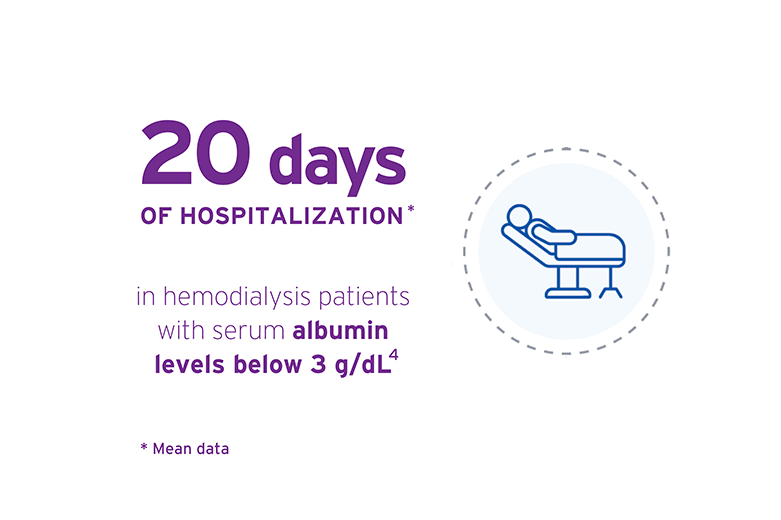
- Kayson GA. Serum albumin concentration in dialysis patients: why does it remain resistant to therapy? Kidney Int. 2003;64 (Suppl 87):592-598.
- Fresenius Medical Care (2016). Instructions for Use: Optiflux High Flux Ebeam F50002272 Rev 07/2023
- Zhou, M., et al. Evaluation of Biomarkers in Chronic Hemodialysis Patients Dialyzed with Optiflux High-Flux Dialyzers, Poster Presentation, NKF Spring Clinical Meetings, 2020. Cause and effect not determined based on design.
- FMCNA data on file as of March 2023.
Indications for use: Optiflux dialyzers are intended for patients with acute or chronic renal failure when conservative therapy is judged to be inadequate.
Caution: Federal (US) law restricts these devices to sale by or on order of a physician.
Note: Read the Instructions for Use for safe and proper use of these devices. For a complete description of hazards, contraindications, side effects, and precautions, see full package labeling at fmcna.com. In rare cases, thrombocytopenia or hypersensitivity reactions including anaphylactic or anaphylactoid reactions to the dialyzer or other elements in the extracorporeal circuit may occur during hemodialysis. The suitability of a dialyzer for a particular treatment is the responsibility of the physician.
© 2024 Fresenius Medical Care. All Rights Reserved. Fresenius Medical Care, the triangle logo, and Optiflux are trademarks of Fresenius Medical Care Holdings, Inc. or its affiliated companies. All other trademarks are the property of their respective owners. P/N 104341-01 Rev C 03/2024