Q: What are the boundary lines?
The Crit-Line profile boundary lines are visual lines displayed on the Crit-Line screen of the 2008T machines, which help to visually represent data related to key threshold rates that have been shown to impact mortality and morbidity outcomes.1
Q: What is the purpose of the boundary lines and how can they be helpful?
They are designed to provide an additional visual aid that supports clinical observation of ultrafiltration tolerance during hemodialysis treatment. When using guidance lines in combination with clinical observation, the blood volume reduction rate zone may help optimize ultrafiltration while reducing risk of intradialytic symptoms.1
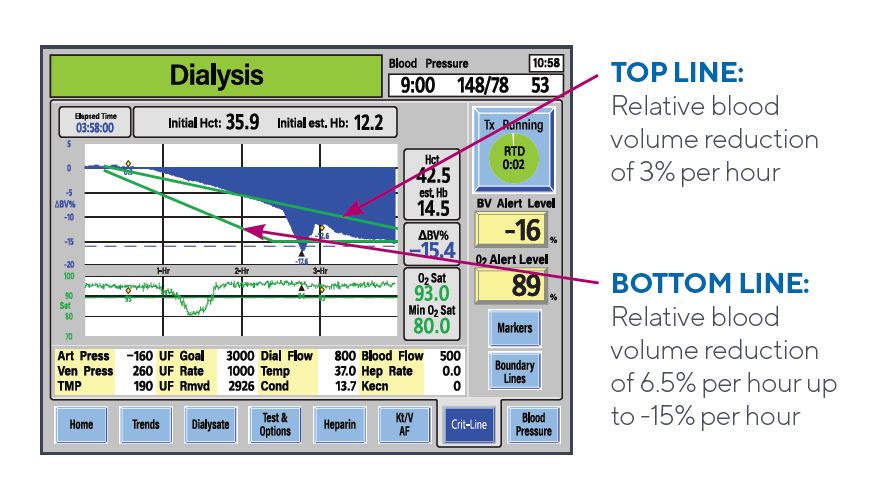
Q: Is there data that shows patients do better when their blood volume profile is between the two boundary lines?
Several publications reference an association between overall reduction of blood volume and improved survivability and reduction of hospitalizations.2,3,4 In 2018, Preciado et al, concluded that specific hourly intradialytic RBV ranges were shown to be associated with lower all-cause mortality in chronic HD patients.1
Q: What does it mean if my patient’s overall blood volume profile is above the top boundary line?
This would indicate the relative blood volume reduction is at or less than 3% per hour, or an “A Profile.“ It is an indication that the patient’s rate of plasma refill is the same as or slightly greater than the rate of ultrafiltration. This suggests the ultrafiltration rate might be increased without immediate risk of intradialytic symptoms if the patient is hemodynamically stable.
Q: What does it mean if my patient’s overall blood volume profile is below the bottom boundary line?
This would indicate the relative blood volume reduction exceeds 6.5% per hour or a “C Profile.” It is an indication of a rapid decrease in blood volume. The rate of ultrafiltration is greater than the rate of plasma refill. Patients in this profile should be observed carefully, as this profile, without any intervention, may bear a higher risk for intradialytic symptoms.
Q: Can I access the original CLiC screen during a patient treatment?
Yes, clinicians can toggle between the profile graphs by touching the on-screen Boundary Line button to add or remove the boundary guidance lines at anytime throughout the treatment.
Q: What software does my 2008T Machine need to have in order to use the boundary lines function?
2008T HD systems equipped with software version 2.65 or higher are eligible for this upgrade.
- Preciado, P, et al, All-cause mortality in relation to changes in relative blood volume during hemodialysis, Nephrol Dial Transplant (2018) 1-8.
- Ficociello LH, Balter P, Taylor PB, Mullen C, Zabetakis PM, Kossmann RJ. Lower Hospital Admission Rate was Associated with Greater Reduction in Relative Blood Volume in Hemodialysis Patients. National Kidney Foundation Spring Clinical Meeting 2015.
- Parker, T. et al., A Quality Initiative. Reducing rates of hospitalizations by objectively monitoring volume removal, Nephrol News Issues. 2013 Mar; 27(3):30-2, 34-6.
- Paul Balter, Paduranga Rao, Yisha Li, Claudy Mullon, Robert Kossmann, Linda Ficociello, Lower Rates of Hospital Admissions During a Fluid Management Quality Improvement (QI) Project Utilizing Relative Blood Volume Monitoring (RBV-M) – A Retrospective Database Analysis, American Society of Nephrology Annual Symposium, 2018.
USE: Crit-Line Clip Monitor (CLiC™) Optional: The CLiC device is used with the 2008T hemodialysis machine to non-invasively measure hematocrit, oxygen saturation, and percent change in blood volume. The CLiC device measures hematocrit, percent change in blood volume, and oxygen saturation in real time for application in the treatment of dialysis patients with the intended purpose of providing a more effective treatment for both the dialysis patient and the clinician. Based on the data that the monitor provides, the clinician/nurse, under physician direction, intervenes (i.e., increases or decreases the rate at which fluid is removed from the blood) in order to remove the maximum amount of fluid from the dialysis patient without the patient experiencing the common complications of dialysis, which include nausea, cramping, and vomiting. The 2008T is indicated for acute and chronic dialysis therapy.
Caution: Federal (U.S.) law restricts these devices to sale by or on the order of a physician.
Note: Read the Instructions for Use for safe and proper use of these devices. For a complete description of hazards, contraindications, side effects, and precautions, see full package labeling at fmcna.com.
© 2022 Fresenius Medical Care. All Rights Reserved. Fresenius Medical Care, the triangle logo, Fresenius Renal Technologies, Crit-Line, CLiC, 2008, and 2008T BlueStar are trademarks of Fresenius Medical Care Holdings, Inc., or its affiliated companies. All other trademarks are the property of their respective owners. P/N 105275-01 Rev A 07/2022
