5 Reasons to Become a Nephrology Clinical Trial Investigator
Clinical trial investigators conduct or supervise clinical investigations that test the safety and efficacy of pharmaceuticals, biotechnology, and medical devices. These practitioners play a key role in gaining regulatory approval for life-saving and life-enhancing therapies that benefit patients everywhere. However, finding enough investigators, while critical, is difficult. Over the past ten years, the number of FDA-registered studies has more than tripled (see Figure 1). This proliferation in clinical trials requires a corresponding increase in physician investigators to drive the research that will improve patients' lives by bringing these novel therapies to market.
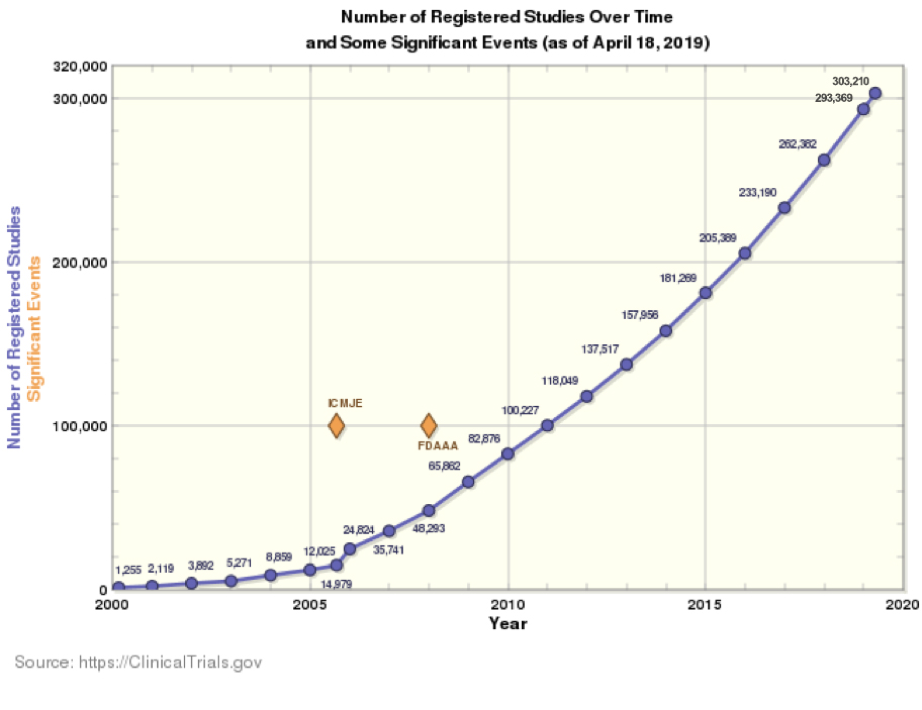
Figure 1.
Why would nephrologists want to offer clinical research? Running a nephrology practice is already a full-time commitment without engaging in study conduct. So why make the effort? Here are five good reasons to start offering clinical research right now:
1. PROGRESS YOUR CAREER
Professional development is a key reason to engage in clinical trials. Participating in cutting-edge research allows principal investigators (PIs) to raise their own professional profiles.
Clinical trial investigators are often seen as thought leaders within their communities. Conducting research may seem particularly noteworthy in nephrology, a field less engaged in research than, say, oncology. Additionally, resume enhancements such as clinical trial conduct and co-authorships in peer-reviewed publications can lead to new professional opportunities.
For personal improvement, by selecting relevant trials, physicians can stay at the forefront of clinical developments in their areas of therapeutic interest. They may also combat feelings of isolation or stagnation by collaborating with other clinical trial investigators. Furthermore, they may gain more personal satisfaction in their career, knowing that their day-to-day work has long-term consequences—that is, that they are making a real contribution to the future of nephrology healthcare.
2. ENHANCE YOUR MEDICAL PRACTICE'S REPUTATION
In addition to the physician's personal and professional advancement, conducting clinical research may also raise the medical practice's profile. Practices that conduct clinical research often tend to be more well known regionally and viewed as more reputable.
Additionally, an apparently progressive and intellectually stimulating environment is likely to attract other nephrologists to the practice, as well as attract and retain more ambitious, eager-to-learn staff. Similarly, the economic opportunities created by clinical research also may draw more talented people into the practice. These considerations are important as physician, nursing, and research assistant shortages in nephrology and other fields continue to climb.1,2
And of course, adding these new staff members enables you to grow your practice, which continues to fuel your office's reputation as a flourishing, forward-thinking practice.
3. GIVE PATIENTS EXTRAORDINARY OPTIONS AND BETTER CARE
Physicians want the best for their patients. Conducting research as part of a nephrology practice allows the practitioner to offer patients therapeutic innovations that are not widely available. Not only do patients gain access to these cutting-edge treatment options that may improve their quality of life, but also the therapy generally comes at little to no cost for patients.
While clinical research may decrease some of the costs of care for the patient and the healthcare system directly, it may also decrease them indirectly. Study patients receive more clinical attention than the average patient. This increase in interaction generally means higher-quality care and increased patient engagement. Studies have shown that patients in clinical studies achieve better outcomes with lower healthcare costs.3,4
4. CREATE A NEW REVENUE STREAM
Pharmaceutical industry trials benefit practices financially by providing an additional, direct source of income. By effectively negotiating reimbursement rates that cover all the per-patient costs, including start-up costs and overhead, physician practices can benefit from increased income.
Indirect benefits—including improved physician and patient engagement, greater physician and practice prestige, and decreased staff turnover—further contribute to the business case for clinical research.5
Furthermore, while kidney disease patients are interested in clinical research studies, they lack access. Patients are attracted to medical practices with a reputation for offering cutting-edge care and having advanced experience with newly approved drugs and devices.
5. ADVANCE GLOBAL HEALTHCARE
Clinical trials do more than improve care for study patients. Conducting clinical trials is a way to make a difference by aiding in the development of new therapies that will improve the future health and well-being of people around the world. In nephrology, the need for research is acute. For complex reasons, fewer randomized controlled trials have been conducted in nephrology than in almost any other discipline (see Figure 2).
Nonetheless, important and meaningful nephrology studies are underway. Given the historically low level of research activity in this field, nephrologists must support these clinical trials if strides are to be made. Only by adding both nephrology research sites and nephrologist-investigators can the pace of kidney research quicken.
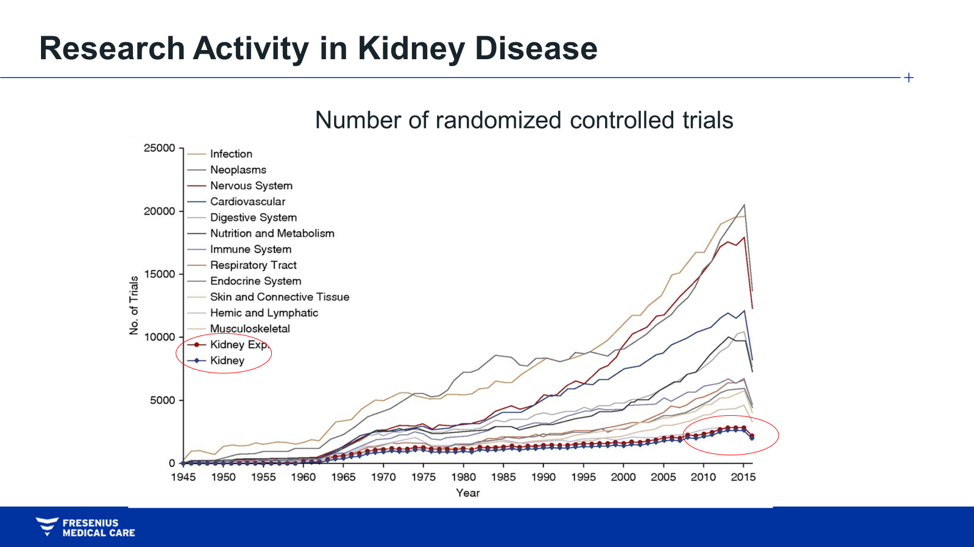
Figure 2. 6
THE TAKEAWAY
Clinical research aids in a physician's personal development and raises both the investigator's and the medical practice's profile while also enabling the investigator to offer newer, more affordable options. Research also provides an additional, direct revenue stream as well as indirect revenue improvements through the attraction and retention of more patients and better staff. For forward-thinking nephrologists and for physicians who want to stimulate their careers, these benefits make clinical research an option worth serious consideration.
References
- Owens, Susan. "The Impending Workforce Crisis in Nephrology." ASN Kidney News. July 2009. Accessed April 22, 2019. https://www.kidneynews.org/kidney-news/special-sections/special-section/the-impending-workforce-crisis-in-nephrology.
- AAMC. “2008 Physician Specialty Data: Center for Workforce Studies.” November 2008. Accessed April 22, 2019. https://www.aamc.org/download/47352/data/specialtydata.pdf.
- Mussina, Kurt, MBA. Advancing Medicine by Routinely Offering Clinical Research to Patents. Report. Frenova Renal Research. 2017. Accessed April 22, 2019. https://www.frenovarenalresearch.com/wp-content/uploads/FMCNA_2017_AMR_Frenova_Kurt_Mussina.pdf.
- Majumdar SR. "Better Outcomes for Patients Treated at Hospitals That Participate in Clinical Trials." Archives of Internal Medicine 168, no. 6 (2008): 657. doi:10.1001/archinternmed.2007.124.
- Song PH, Reiter KL, Weiner BJ, Minasian L, McAlearney AS. "The Business Case for Provider Participation in Clinical Trials Research." Health Care Management Review 38, no. 4 (2013): 284-94. doi:10.1097/hmr.0b013e31827292fc.
- Kyriakos M, et al, “Quantity and reporting quality of kidney research.” JASN, no. 30 (2019):13-22.



